One of the main reasons I started this blog and have decided to continue posting is my simple yet strong belief that objectivity and truth are essential when both analyzing and discussing political or public health decisions. I have been thinking about writing this post for over a year and, unfortunately, there is still a lot of misinformation and lack of understanding regarding COVID-19 in the general public as the virus continues to rage in our country and the world at large.
For this post, I researched questions that are commonly asked about COVID-19 and provided a referenced summary of what I learned. Quick and simple answers to each question are provided at the top of each section followed by a deeper dive. If you have further questions please comment and I will do my best to respond with credible information.
Thank you for reading!
COVID-19 FAQs:
- What is COVID-19?
- Where did COVID-19 come from?
- Isn’t COVID-19 like the flu?
- Do masks and social distancing work?
- Would a nation-wide shutdown help?
- What happens if I get COVID-19?
- Do COVID-19 tests work?
- Are the vaccines safe?
- What does the future look like?
Some Quick Definitions:
- Virus: microscopic pathogens unable to replicate on their own, requiring the infection of host cells for replication and existence.
- Viral Classification System: viruses follow the same taxonomic ranks as living organisms: domain, kingdom, phylum, class, order, family (there can be sub families as well), genus, and species, but some commonly used terms come up beyond the species level:
- Strain/Type/Variant: essentially all mean the same thing, but are used within different naming conventions – all refer to a virus with a genetic and/or phenotypic feature setting it apart from other viruses within the same species.
- Genome & Genetics: the genome refers to the entirety of an organism’s genetic information. Genetics refers to the information encoded in nucleic acids (DNA & RNA).
- Phenotype: the observable features of an organism, encoded by nucleic acids,
- Antigens & Antibodies: antigens are foreign entities within the cell (produced by viruses or other infectious agents) which cause the immune system to create antibodies. Antibodies are proteins produced by the immune system that are engineered to target and ultimately rid the cell of a specific antigen, clearing out pathogens.
1. What is COVID-19?
COVID-19 is the disease caused by SARS-CoV-2, a new type of coronavirus that emerged in 2019. COVID-19 stands for “Corona Virus Disease 2019” and SARS-CoV-2 stands for “Severe Acute Respiratory Coronavirus 2”.
Although we are dealing with a novel species of coronavirus we are not dealing with a novel type of virus. In fact, there are hundreds of known coronaviruses; six were already known to infect humans before SARS-CoV-2, including the previous species to cause severe acute respiratory syndrome (SARS) in 20021.
Coronaviruses, or Coronaviridae, are a family of membrane-enveloped, positive-sense RNA viruses. Coronaviruses (CoVs) were first detected in humans in the 1960s, but most species can be found in animals ranging from domesticated livestock to wildlife2. All coronaviruses share a similar outer cell membrane structure with spikes that resemble a corona, or “crown” (image 1). Within the family, coronaviruses are further categorized by uniquely shared genomic and phenotypic features into four genera: Alpha, Beta, Gamma, and Delta. SARS-CoV-2 and SARS-CoV are both Betacoronaviruses3.
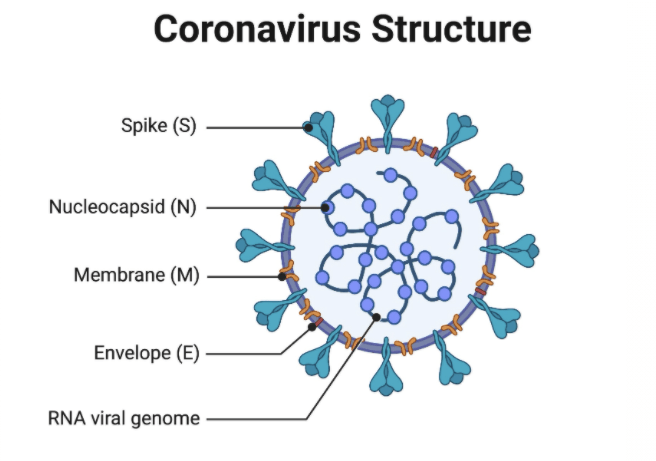
(source: https://www.biophysics.org/blog/coronavirus-structure-vaccine-and-therapy-development).
Including this most recent virus there are now seven coronavirus species that can infect humans (image 2) causing mild to severe respiratory illnesses ranging from the common cold to SARS and MERS, Middle East Respiratory Syndrome, both of which can be fatal4. All seven of the known species of human coronaviruses can be traced to animal reservoirs, meaning that certain animal species are known carriers of the viruses. These animal carriers are often asymptomatic and do not show any adverse effects themselves, but are able to readily spread the virus to new hosts.

Why is the virus called SARS-CoV-2 and what does COVID-19 stand for?
The coronavirus that caused the 2002 SARS outbreak in the Guangdong province of China was named SARS-CoV because it was the SARS-causing coronavirus. This new species responsible for the current pandemic is likewise titled SARS-CoV-2, because it is the second species to cause SARS in humans (“Second SARS-Causing Coronavirus”). COVID-19 is the name of the disease caused by SARS-CoV-2 and stands for “Corona Virus Disease 2019”.
What makes SARS-CoV-2 different from SARS-CoV?
Its exact genetic code has variations, or differences, within it that set it apart from all other known coronaviruses, including SARS-CoV. The genetic code, or sequence, of the new virus was identified once it was isolated from patients with the disease we now call COVID-19. Once the sequence was identified it could be compared to other coronavirus species, allowing us to see how closely related it is to others. SARS-CoV is the most closely related human coronavirus, but other coronaviruses found in animals, and more specifically bats, are also closely related. The relativity between virus species is used to determine where a virus originated from and how it crossed over to infect humans from animals.
2. Where did COVID-19 come from?
The exact origin of SARS-CoV-2 is still being determined, but all previous human coronaviruses have originated from animal hosts. Horseshoe bats are carriers of coronavirus species very similar to SARS-CoV-2 and the virus most likely originated from a bat carrier, though it is still being determined if there was an intermediate host between bats and humans.
Viruses are not considered living beings because they lack the ability to reproduce on their own – one of the seven characteristics of life (for a refresher on viruses and other microbes, please visit my previous post). Instead, viruses are essentially capsules containing genetic information, or what could be considered a viral genome, that can transport and spread this information from host to host and/or cell to cell. To “reproduce”, or replicate, a virus must insert its genetic information into a host’s cell, and the host’s own cells will assist with the replication of viral particles. As more viruses are created within the cell, it will eventually die and release viruses to neighboring cells. This is the basis for infection and disease caused by viruses within the host.
When inside a host cell, a virus’ genome can mutate either through a change in its own genetic code, or by “mixing” genetic information with another virus. This is a big oversimplification but the general ideas are that an error can occur during the replication process, leading to a change in the genetic code, or alternatively, if two or more viruses happen to co-exist within a cell for long enough they can recombine their genetic information during replication. Either process can result in functional changes for the virus – including the ability to infect a new type of species. When a virus is able to “jump” from an animal species into humans it’s considered a zoonotic spread and the disease it causes is a zoonotic disease.
Sometimes you will hear people describe a virus as “evolving”. This essentially means that the virus’ genome has changed, or mutated, to the point that they are considered a novel type of virus – they can still be classified by the group of viruses they are related to and evolved from, but they have unique genetic codes and characteristics that now set them apart. When a virus has changed enough that it can infect a new species, jumping from an animal to humans for example, it is generally said that the virus “evolved to infect humans”.
Some viruses evolve so rapidly that new strains are frequently created or co-exist within a population, leading to the need to continuously update vaccinations and treatments. This is the case with influenza, the cause of the common flu. Each year the flu vaccine is adjusted to fight against what is believed to be the most common flu strain that season. Sometimes a different strain is actually the most infectious, or a new mutation leads to the vaccine being less effective, so the flu vaccine doesn’t always get it right; it does, however, prevent millions of infections each year5 and medical professionals generally recommend getting the vaccine each flu season.
These “ever evolving” viruses will most likely indefinitely remain in the human population due to their ability to adapt to therapies, and viruses will continue to evolve to jump from animals to humans due to their continual genetic updates.
As mentioned before, all seven of the coronaviruses in humans can be traced to animal reservoirs, and most can be traced to the exact species that directly acted as the donor to human hosts6. Zoonotic spread of viruses usually occurs through the consumption of animal meat, though it can also happen from simply being in close proximity to animals, such as the conditions found in live animal markets or factory farms7.
We know zoonotic spread occurs because of each virus’ genetic similarities. We now have the ability to sequence, or read, the entire genome of viruses and compare them. The genome is mind-blowingly rich with information, or data. Through genomic data, we can determine if a virus is related to another and how closely they are related – this is an essential step for identifying the cause of a viral outbreak. The genome also contains information on how the virus spreads and infects human cells, helping us to further isolate the cause as well as formulate treatments.
The SARS-CoV outbreak in 2002 was traced back to Masked palm civets (Paguma larvata), or “racoon dogs”, in a live animal market. The genome of the coronavirus isolated from humans infected with SARS was almost identical to the genome of the coronavirus isolated from civets found in the market8. This led to the order to kill all the civets, shortly after which the outbreak ended, further suggesting that the zoonotic spread had ended with the civets (cruel) removal. However, all the animal species in the market were tested for antibodies against SARS-CoV, which if positive would suggest that they had at one point and most likely recently been infected with the virus. Antibodies were found in 80% of the different animals8.
Included in this antibody-positive group were Chinese horseshoe bats (Rhinolophus sinicus) which were found to contain a coronavirus very similar to SARS-CoV in humans8. In fact, bats have been identified as the main animal reservoir for all human coronaviruses9. A coronavirus isolated from horseshoe bats in the early 2000s shares 96.2% nucleotide sequence homology with SARS-CoV, meaning that 96.2% of their genomes match and suggests that the human virus originated from the bat virus.
Similar to SARS-CoV, a coronavirus isolated from Intermediate horseshoe bats (Rhinolophus affinis) shares 96% homology with the SARS-CoV-2 genome10. It has yet to be determined if the human virus jumped directly from bats to humans or if another animal acted as an intermediate host, but all evidence points to COVID-19 being a zoonotic disease. It has not been determined that the cause of the SARS-CoV-2 outbreak was the wet market in Wuhan, China, but the spike in spread and subsequent environmental testing suggests that there were infected animal hosts present11.
How do we know it wasn’t engineered in a lab?
As described before, genetics is like data and those trained to understand that data can determine if there have been “man-made alterations” because it would leave behind telltale signs. When humans perform genetic engineering there are a very small number of tools that can be used, and each leaves it’s “mark” on the genome which can then be detected, usually by the presence of genetic sequences, or codes, that naturally should not exist at the number or location in which they do in a genetically modified genome. I recommend this article if you’d like to read more about the evidence against COVID-19 being a lab-engineered virus.
3. Isn’t COVID-19 like the flu?
No. COVID-19 and the flu are both respiratory illnesses that have some shared symptoms, but COVID-19 is more infectious, causes more severe symptoms, and has a higher mortality rate compared to the flu.
Beyond both causing respiratory illnesses, influenza viruses and coronaviruses are quite different, just as the flu and COVID-19 share some symptoms but are very different illnesses.
There are four influenza viruses, belonging to the Orthomyxoviridae family and Orthomyxovirus genus – influenza A, B, C, and D. Influenzas A & B both cause the seasonal flu, and influenza A was responsible for the Spanish flu pandemic. Differences in the flu virus structure, among other variations, lead to different mechanisms of entering host cells and replicating within the cell (figure 3) compared to coronaviruses, which is why COVID-19 and the flu infect people at different rates and with different effects.
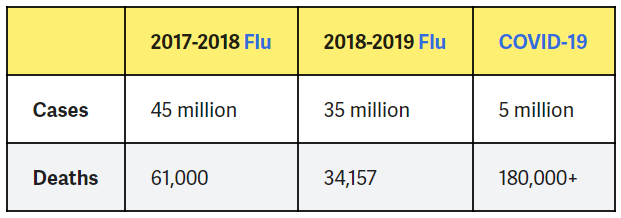
Since the pandemic is ongoing the rate of spread, otherwise called the contagion or infection rate, and death rate are still being determined, but so far by looking across patients with COVID-19 and patients with the flu (both currently and historically) COVID-19 has a significantly higher rate of spread, and has a significantly higher death rate, along with showing signs of causing chronic diseases not associated with the flu12. Previous to COVID-19, the biggest pandemic humans experienced was the Spanish Flu (1918-1920); it’s estimated that 500 million, or 1/3 of the world population, became infected and out of those infected 50 million died, including 675,000 Americans13. As of January 11, 2021 an estimated 90.4 million people have been infected by COVID-19 worldwide, with almost 2 million deaths, including nearly 375,000 Americans14.
The common flu generally leads to 30,000-40,000 US deaths per year or up to 650,000 globally and has a mortality rate of about 1% or less15. In striking contrast, the mortality rate of COVID-19 is as high as 29% in countries hit hardest and is about 2% in the United States16. The difference in country-specific mortality rates is due to a number of variables17:
- Access to COVID testing: testing can prevent deaths by prompting those who test positive to get healthcare early on.
- Access to proper healthcare: those that require medical attention need the ability to both get to the doctor and to also get the proper treatments.
- Culture: cultures that already practice or are more willing to practice social distancing and wearing masks generally have lower rates of mortality.
- Socioeconomics: countries with higher rates of poverty are generally experiencing higher mortality rates.
- Demographics: countries with higher populations of those 65 and over are generally experiencing higher mortality rates than others; also populations that have a higher rate of comorbidities (cancer, heart disease, chronic respiratory disease, etc.) have higher rates of mortality. Air quality and higher rates of smoking are also linked to increased mortality rates.
Minority groups have generally been hit hardest by SARS-CoV-2, with higher rates of infection and mortality compared with white individuals in the same areas. Though there may be some ethnicity-based genetics that influence this, research has identified factors to be socioeconomics and due to cultural biases. Minorities are more likely to work in a position that puts them at risk of exposure, while also being less likely to receive proper medical care, often due to prejudices within healthcare, and on the other side fear of seeing doctors due to past discrimination. Chronic stress can also increase the risk of getting COVID-19 and of having a more severe reaction, and it has long been proven that minorities often endure chronic stress due to racism they experience18.
A study released in December 2020 looking at COVID-19 and flu patients within a French hospital found that almost twice as many people were admitted to the hospital with COVID-19 than the flu over a 2-month period when flu season occurs19. Of these hospitalizations, respiratory complications were more common in COVID-19 patients, who were twice as likely to end up in the ICU. More significantly, the in-hospital mortality of COVID-19 patients was three-times higher than those with the seasonal flu, and still significantly higher than the worst seasons of influenza the hospital had experienced in the past 5 years.
Evidence shows that SARS-CoV-2 is more infectious than influenza thus spreading through the population more quickly and when infected, COVID-19 causes more serious respiratory issues and has a significantly higher probability of being fatal when compared with the flu.

4. Do masks and social distancing work?
Yes. SARS-CoV-2 is spread through respiratory droplets – masks and social distancing reduce the likelihood of droplets coming into contact with your own respiratory system, preventing infection.
SARS-CoV-2 is a respiratory virus, meaning that it specifically infects cells within the respiratory system. Humans will secrete the virus in respiratory droplets – saliva or other secretions. No matter how hard we try, we release these respiratory droplets into the air around us; even more so when we are talking or eating. To be infected with SARS-CoV-2, you either come in physical contact with a person or surface where a virus-carrying respiratory droplet exists (contact transmission) or you breathe in a respiratory droplet from the air (droplet transmission). Thankfully SARS-CoV-2 cannot survive for long outside of host cells, so it’s not capable of airborne transmission and contact transmission from surfaces is relatively rare. Most SARS-CoV-2 transmission comes from breathing in respiratory droplets from a nearby person.
Social distancing helps lower the transmission rate significantly by reducing the likelihood someone will breathe in respiratory droplets from a SARS-CoV-2 carrying person. Previous studies have found that most droplet transmission occurs when people are within three feet (1 meter) of each other20 and sneezes can cause droplets to travel 6 feet (2 meter) and beyond. This is why guidelines tell us to keep at least 6 feet away from each other – so you don’t breathe in infected droplets, but droplets can travel further than 6 feet so it’s best to keep as far away from others as possible during these times.
When social distancing is not possible, and even with the six foot rule can be followed, masks help to further reduce the transmission of SARS-CoV-2 by acting as a shield against the respiratory droplets. It’s true that the viruses are too small to be shielded by most masks, though some filters are efficient at removing these as wells, but the viruses are not entering the respiratory system alone. Instead, they have to hitch a ride in droplets, so if we can prevent them from entering the mouth or nose, the virus will also be blocked.
Surgical and cloth masks, like the ones worn by healthcare professionals and the variety of masks now available to the public, are efficient at blocking most droplets, especially larger ones (>10 um) – if worn properly (figure 4). Smaller droplets (<2.5 um) may still get through the masks; N95 masks are better are blocking these smaller droplets, but are not always 100% efficient – this is why both social distancing and masks are required to end transmission.
Masks and social distancing have proven to be effective when followed by a community. Studies have shown that when mask ordinances have been put in place on state or local levels, the COVID-19 infection rate dropped up to 2% per day21. To help put this in perspective, epidemiology guidelines dictate that a COVID-19 positive rate below 5% within a community for at least 2 weeks is considered the threshold under which businesses can open up fully. This is because the transmission rate is then manageable and population-wide outbreaks are not likely to occur22. Currently most US states are well above this 5% threshold and COVID-positive rates are reaching high double-digits, with the average across the US currently around 12%23.
If a state is experiencing a 15% positivity rate, enforcing a mask mandate along with social distancing, including the temporary shutdown of in-person and non-essential businesses, could drop the positivity rate to below 5% potentially within one to two weeks. Following this, businesses could open back up without fear of further major outbreaks and repeated shutdowns. However, for these measures to work, everyone has to participate.
Groups of people protesting the social distancing rules, not wearing masks in public or reducing their in-person interactions are causing the pandemic to spread and continue to devastate the country; it is because of these actions that businesses cannot open fully. So if you want businesses to open and the economy to recover – promote the use of masks and following social distancing guidelines.

Could wearing a mask caused me to suffer loss of oxygen or cause any long-term negative effects?
Simply put, there is no evidence that masks cause depletion of oxygen or lower your oxygen levels, nor will they trap carbon dioxide when you breathe out. Masks have not been linked to causing any health disorders, including cancer. However, masks can cause disruptions in breathing patterns and you should only be using certain fabrics to be at your safest.
Masks can cause us to actually breathe differently, often subconsciously, so what feels like the mask causing us to not get enough oxygen, is our reaction to the mask causing us to either hyperventilate – breathe too quickly – or hypoventilate – breathe too slowly24. If you have a mask that feels like it’s “choking” you, the material or fit is most likely to blame. Cloth masks are recommended, specifically those with two layers, but even a single layer should be sufficient with proper social distancing; medical masks (surgical and N95) can also be used, though N95 masks require proper fitting and the CDC has recommended that N95 masks be reserved for healthcare workers25. Masks should have a seal around the nose and mask, but it does not need to be so tight that it causes an imprint or feels restrictive.
There are rare respiratory conditions that may prevent someone from wearing a mask26, however those with these conditions are also at a higher risk of contracting a severe form of SARS-CoV-2 and so should be practicing stay-at-home measures as strictly as possible for their safety.
5. Would a nation-wide shut down help?
It would help reduce the rate of new transmission significantly, helping hospitals deal with the recent influx of cases and preventing a number of deaths. However, at this stage we are most likely at such a high number of cases that it would not prevent future outbreaks from occurring, and we cannot “flatten the curve” without vaccinations.
A total shutdown, or “lockdown”, where everyone stays in their homes for a period of time, most likely 10-14 days for this virus, should greatly assist with reducing transmission rates and effectively flattening a virus’ transmission curve – meaning that further mass spread, or outbreak, is prevented27.
This is because those who are infected with COVID-19 at the beginning of the lockdown cannot spread the virus to others and can receive treatment; and because new infections are not occurring, hospitals can more effectively treat these patients. Throughout the lockdown period new transmission should be near zero (essential work will still occur, so the possibility of continued transmission is still there but much lower), and those with long-term symptoms should be in treatment as well as remain quarantined after the shutdown has ended. This brings the total number of active cases down substantially.
Once the lockdown is lifted, masks and social distancing should be observed, but person-to-person transmission will begin again. However, the amount of people with infections should be low enough that the rate of transmission will be manageable, and outbreaks are prevented.
Unfortunately, shutdowns are only effective if everyone does them, so most measures taken by governments and businesses so far have largely been in vain. With the current number of infections in the United States, a lockdown may not reduce the transmission rate to one that is manageable, and mass spread is most likely only preventable with the introduction of vaccines. However, a lockdown would still assist with lowering the transmission rate and helping lower hospital capacity.
Currently, most states have over 75% of their ICU beds occupied, with many states reaching full capacity28. This means that not only are those with COVID-19 in risk of not being able to get medical care when it is much needed, anyone with a medical emergency may need to be turned away. Prior to the pandemic, ICU capacity across the states averaged 68% and there had been no significant increase in this number during influenza season29.
6. What happens if I get COVID-19?
The most likely symptoms are similar to the cold and flu, but in some cases you will require hospitalization, a stay in the ICU and potentially face death.
If you are infected with SARS-CoV-2 and get COVID-19, your symptoms will typically begin to occur 2-14 days after initial infection. Symptoms range from mild to severe and include fever, chills, coughing, difficulty breathing, muscle aches, fatigue, nausea, and diarrhea, among others30. Most people with COVID-19, even those with mild symptoms, have reported a loss of taste or smell, and in some of these cases the loss can be long-term31. Thankfully, most COVID-19 cases do not require hospitalization, but if someone has an underlying condition, such as hypertension, diabetes, or cardiovascular disease their likelihood of having severe symptoms and requiring hospitalization is greatly increased32.
If you are unfortunate enough to have severe symptoms, your doctor may prescribe you Bamlanivimab, a new FDA-authorized therapy using monoclonal antibodies to help the immune system recognize the virus and respond effectively33. If your symptoms are so severe that you require hospitalization, there are currently no 100% effective cures for COVID-19, meaning a treatment that will kill the virus directly and completely, so doctors mostly concentrate on fighting the symptoms and ensuring vital organs function properly. If you are having difficulty breathing, you may require a ventilator or a nasal cannula to deliver oxygen, and if you lose the ability to eat you’ll have nutrients delivered via an IV.
There are rare cases of COVID-19 as well that cause issues ranging from neurological problems to blood clots. Some patients have developed life-altering neurological disorders including seizures and psychosis34. Other patients have had to have limbs and extremities, such as legs and hands, amputated due to blood clots formed from complications with COVID-1935. Unfortunately, there will continue to be rare and potentially novel cases as each person’s immune response and viral infection are unique.
So far, the only COVID-19 specific treatment that has been administered within general hospitals is Remdesivir, an anti-viral medication made by Gilead Sciences that prevents SARS-CoV-2 from replicating. However, another treatment has just recently been granted FDA Emergency Use Authorization (EUA) – Regeneron’s REGN-COV2, which is an antibody treatment like Bamlanivimab, but is reserved for more severe cases. These antibody treatments deliver antibodies that will act like the cell’s own immune response, both recognizing and neutralizing SARS-CoV-2 antigens. Both antibody treatments have been largely unavailable to the general public, but should be available throughout the country by 2021.
Although the probability of having a severe reaction to COVID-19 is low if you are healthy and young, you may spread the virus to someone with a compromised immune system, such as someone dealing with cancer or a chronic illness, or those in an older age bracket, and these people could end up in the ICU or dead. It’s each of our responsibilities to care for those outside of ourselves, and prevent the virus from spreading.
7. Do COVID-19 tests work?
Yes, but they have their limitations. In general, trust positive results and confirm negative results. If you want to know if you currently have COVID-19, make sure you get a diagnostic test and not an antibody one.
There are two general types of testing for COVID-19: diagnostic and antibody testing. Diagnostic testing is looking for the virus itself, either through the detection of a genetic code unique to the virus or through the detection of a viral antigen, such as a protein, again unique to the virus. Antibody testing is looking for COVID-19 specific antibodies which we create as part of the immune response to the virus’ presence. Antibody testing may or may not tell you if you currently have COVID-19 since antibodies can take up to 1-3 weeks after initial infection occurs36. Instead, antibody tests are meant to detect past infections.
There have been hopes that being infected by COVID-19 leads to temporary or indefinite immunity, due to the presence of antibodies, which have been detected up to 8 months after infection37, however there is mixed evidence on antibodies always leading to immunity, and still nothing substantiating that once infected you can’t get re-infected; in fact several people have been re-infected within months of their initial infection38.
Diagnostic tests are intended to check for current infection, and most of those created for COVID-19 are Real-Time (RT) PCR-based, which means that the tests are looking for a single or several unique genetic codes specific to SARS-CoV-2 through RT-PCR technology. Other tests are looking for SARS-CoV-2 specific proteins. These genetic codes or proteins are detected within a host’s sample, usually mucous from a nasal swab or saliva from a cheek swab.
Why do people get false positive or false negative results?
False positive results can occur when the specificity of the test is off and the test believes it has identified a viral gene or protein that is actually the host’s. This occurs because humans contain genetic codes that can be very similar to the virus’s, and our protein’s structures can also be closely related to viral ones, “tricking” the test into thinking what it detects is viral because it does not have a high enough degree of specificity to tell the difference. On the other hand, false negative results can occur when either the viral material within the given sample is in such a low amount that it cannot be detected or it happened to be not be captured in that sample at all; this is more common in people who are newly infected and still pre-symptomatic and those that remain asymptomatic. Technical errors with tests can also contribute to inaccurate results39.
If you’ve been around someone with COVID-19 it’s best to quarantine and wait at least 48-72 hours before getting a diagnostic test to ensure the virus can be detected if present, and if the test is negative it’s recommended to get a second test 2-3 days later to confirm. Quarantining for 10-14 days after exposure should always occur regardless of the results to ensure the virus isn’t spread when people are asymptomatic.
8. Are the vaccines safe?
Yes. Some people will have allergic reactions, but these do not cause long-term effects.
There are two FDA-authorized vaccines currently available for SARS-CoV-2: Pfizer-BioNTech’s COVID-19 Vaccine (BNT162b2) and Moderna’s COVID-19 Vaccine (mRNA-1273), both of which are mRNA vaccines. In general, vaccines create immunity against a pathogen by causing a natural immune response within the body specific to that pathogen, in a safe manner – meaning that an inactive form or particle of the pathogen, in this case SARS-CoV-2, is introduced to the body, eliciting an immune response which creates antibodies against that virus, so if the pathogen does infect a cell, it is quickly recognized and destroyed via those “pre-made” antibodies.
The Central Dogma of genetics is the process of converting the information in DNA into proteins. You can think of RNA as the messenger in between DNA and proteins, which is where we get the name messenger RNA, or mRNA. DNA is converted into mRNA during a process called transcription, and the mRNA is then used to create proteins during a sequential process called translation.
With mRNA vaccines, an immune response is elicited by the introduction of mRNA strands which encode partial viral proteins, or antigens, that the body will produce antibodies against. When the mRNA is introduced into one’s cells, naturally-occurring translation will lead to the production of the protein the mRNA encodes for. Both the Pfizer-BioNTech and Moderna vaccines use mRNA that produce partial SARS-CoV-2 membrane spike proteins, acting as antigens and leading to an immune response and the production of antibodies that target those specific proteins. If SARS-CoV-2 infects you after being vaccinated, the body will readily recognize the virus by this protein and can destroy the virus before it can cause illness.
Although there has been no evidence that vaccines cause serious illness or adverse effects in the general population, mRNA vaccines are considered even safer than traditional vaccines because only a single viral protein or partial protein is introduced into the body and mRNA breaks down more rapidly than larger components within cells, removing it more quickly from cells and reducing the likelihood of any negative effects being triggered40.
It may be a concern to have mRNA introduced into cells which encodes something viral, however mRNA does not have the ability to incorporate or interact with our DNA, causing no effects to our own genetics. Once mRNA is used to create a protein, the cell will degrade the mRNA, so it cannot be “re-used”41. The only effect on our cells will be the production of partial viral proteins, which have been proven to have no negative effect on people during clinical trials42. Both the Moderna and Pfizer-BioNTech vaccines provide doses of mRNA that lead to a relatively low amount of viral particles being made, so cells will not be overwhelmed while still able to elicit an immune response. Allergic reactions can occur in rare cases however.
Just as there will be different reactions to SARS-CoV-2 or the flu, there are different reactions to vaccinations. Some people’s bodies may respond strongly even to partial viral proteins, which can lead to inflammation and other symptoms that can be detrimental if they are too strong or persist for too long. The few severe allergic reactions to the COVID-19 vaccines so far have been in individuals with other strong allergies, suggesting that the body was sent into an “immune response overdrive” with the elicitation of a new immune response. The CDC recommends that those with severe allergies discuss these with their doctor prior to vaccination to determine if the risk is too high43.
For those without allergies, you may still feel some short-term side effects following the vaccination. This is because the vaccine is causing an immune response, which can lead to side effects such as pain and swelling at the shot site, as well as fevers or chills, migraines, and fatigue44. In general, vaccines produce a severe allergic reaction every 1 in 1-million doses45. The rate of allergic reactions against the two mRNA vaccines are still being determined, but as of 250,000 doses, 6 allergic reactions had been reported which is a rate of 0.024%46.
How many people need to be vaccinated before we have ‘herd immunity’?
In the case of SARS-CoV-2, its estimated that 75% of the population needs to acquire immunity to the virus before the rate of transmission is considered near zero, or halted47, at this point it would be said that the population has “herd immunity” to the virus. Since there is still conflicting evidence that being infected with SARS-CoV-2 leads to long-term immunity, this essentially means that 75% of the population needs to be vaccinated before business can return to normal. Currently only about 0.9% of the population has been vaccinated as of December 31, 202048.
Will we need new vaccinations each year, like with the flu?
As mentioned before, the reason that the flu vaccine has to be updated each year is because the influenza virus mutates, or evolves into new types, rapidly – creating new ways to evade the immune response previously crafted against the virus, and effectively making a previous vaccine ineffective. The coronavirus similarly has several different strains, all most likely originating from a single one; this suggests that the virus will continue to evolve. Thankfully, the rate of change and ways in which the virus is changing do not seem significant enough to cause alarm with current vaccine effectiveness49. It’s believed that both mRNA vaccines will be effective against all current SARS-CoV-2 strains, but it may be that these vaccines will also need to be updated overtime.
9. What does the future look like?
As bright as we make it.
The future is still very much to be determined, but there is a light at the end of the dark tunnel the pandemic has caused. Vaccinations are rolling out globally, treatments are still being discovered and improved, and people across the world are working tirelessly to help prevent further illness.
There should be specific appreciation shown to public health workers, frontline healthcare workers, scientists, teachers, essential food workers, and everyone else who is ensuring people are fed and cared for during these times.
There are still a lot of painful and hard months ahead that we must get through. Until vaccines have led to a high enough rate of immunity across populations, people will continue to get sick and die, and hospitals will continue to be overran when outbreaks occur. We must follow public safety measures including wearing masks and social distancing until we can call the virus defeated, and even after that we must stay vigilant and potentially continue to update our vaccines and treatments as the virus evolves.
We must also think beyond COVID-19. As much as we would like it to be, this will not be the last pandemic to sweep across the country or globe, so we must also continue to educate ourselves and promote measures that ensure public safety.
Thank you again for reading, and please keep each other safe.
Special thank you to Ryan Pierce for editing.
References:
- https://www.healthline.com/health/coronavirus-types
- https://pubmed.ncbi.nlm.nih.gov/16378050/
- https://www.sciencedirect.com/topics/biochemistry-genetics-and-molecular-biology/coronavirinae
- https://www.niaid.nih.gov/diseases-conditions/covid-19
- https://www.cdc.gov/flu/prevent/keyfacts.htm#:~:text=Flu%20vaccination%20can%20keep%20you%20from%20getting%20sick%20with%20flu.&text=For%20example%2C%20during%202019%2D2020,and%206%2C300%20influenza%2Dassociated%20deaths.
- https://www.nature.com/articles/s41579-018-0118-9
- https://www.pnas.org/content/pnas/110/21/8399.full.pdf
- https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7098031/
- https://virologyj.biomedcentral.com/track/pdf/10.1186/s12985-015-0422-1.pdf
- https://wwwnc.cdc.gov/eid/article/26/7/20-0092_article
- https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7086482/
- https://www.meaction.net/2020/05/10/researchers-expect-covid-19-will-cause-surge-of-chronic-illness-including-me/
- https://www.cdc.gov/flu/pandemic-resources/1918-pandemic-h1n1.html
- https://www.cnn.com/interactive/2020/health/coronavirus-maps-and-cases/
- https://www.who.int/news/item/11-03-2019-who-launches-new-global-influenza-strategy
- https://coronavirus.jhu.edu/data/mortality
- https://www.nature.com/articles/s41598-020-75848-2
- https://www.npr.org/sections/health-shots/2020/04/18/835563340/whos-hit-hardest-by-covid-19-why-obesity-stress-and-race-all-matter
- https://www.thelancet.com/journals/lanres/article/PIIS2213-2600(20)30527-0/fulltext
- https://www.who.int/news-room/commentaries/detail/transmission-of-sars-cov-2-implications-for-infection-prevention-precautions
- https://www.healthaffairs.org/doi/10.1377/hlthaff.2020.00818
- https://www.jhsph.edu/covid-19/articles/covid-19-testing-understanding-the-percent-positive.html
- https://coronavirus.jhu.edu/testing/individual-states
- https://www.discovermagazine.com/health/why-it-feels-like-you-cant-breathe-inside-your-face-mask-and-what-to-do
- https://www.healthline.com/health-news/best-materials-for-covid19-face-masks
- https://www.technologynetworks.com/immunology/news/exemptions-of-respiratory-patients-to-use-face-masks-are-not-evidence-based-341164
- https://www.cdc.gov/coronavirus/2019-ncov/if-you-are-sick/quarantine.html
- https://healthdata.gov/dataset/covid-19-estimated-patient-impact-and-hospital-capacity-state/resource/82e733c6-7baa-4c65#{view-graph:{graphOptions:{hooks:{processOffset:{},bindEvents:{}}}},graphOptions:{hooks:{processOffset:{},bindEvents:{}}}}
- https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3840149/
- https://www.cdc.gov/coronavirus/2019-ncov/symptoms-testing/symptoms.html
- https://www.webmd.com/lung/news/20200604/covid19-loss-of-smell-taste-might-be-long-term#1
- https://www.cdc.gov/mmwr/volumes/69/wr/mm6915e3.html
- https://www.cdc.gov/coronavirus/2019-ncov/your-health/treatments-for-severe-illness.html
- https://www.nature.com/articles/d41586-020-02599-5
- https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7188654/
- https://www.cdc.gov/coronavirus/2019-ncov/testing/serology-overview.html
- https://www.cidrap.umn.edu/news-perspective/2020/12/two-studies-find-covid-19-antibodies-last-8-months
- https://www.sciencemag.org/news/2020/11/more-people-are-getting-covid-19-twice-suggesting-immunity-wanes-quickly-some#:~:text=Since%20then%2C%20at%20least%2024,symptom%2Dfree%20month%20in%20between.
- https://health.ucdavis.edu/health-news/newsroom/different-types-of-covid-19-tests-explained/2020/11
- https://www.hopkinsmedicine.org/health/conditions-and-diseases/coronavirus/is-the-covid19-vaccine-safe
- https://www.cdc.gov/coronavirus/2019-ncov/vaccines/different-vaccines/mrna.html#:~:text=A%20Closer%20Look%20at%20How%20COVID%2D19%20mRNA%20Vaccines%20Work&text=COVID%2D19%20mRNA%20vaccines%20are,and%20gets%20rid%20of%20them.
- https://www.pbs.org/wgbh/nova/article/pfizer-moderna-covid-vaccines-clinical-trials/
- https://www.cdc.gov/coronavirus/2019-ncov/vaccines/safety/allergic-reaction.html
- https://www.cdc.gov/coronavirus/2019-ncov/vaccines/expect/after.html
- https://www.entnet.org/content/allergic-reactions-related-covid-19-vaccinations-allergic-patients
- https://www.wcvb.com/article/allergic-reactions-250k-covid-vaccine-shots/35021120
- https://www.statnews.com/2020/12/17/calculating-our-way-to-herd-immunity/
- https://www.axios.com/coronavirus-vaccines-administered-2020-9ec0cebf-58cd-4294-8b1e-9334c76363c8.html
- https://today.tamu.edu/2020/12/22/texas-am-expert-vaccines-should-control-new-covid-19-strain/
